Ideal Tips About How To Find Out Electrons

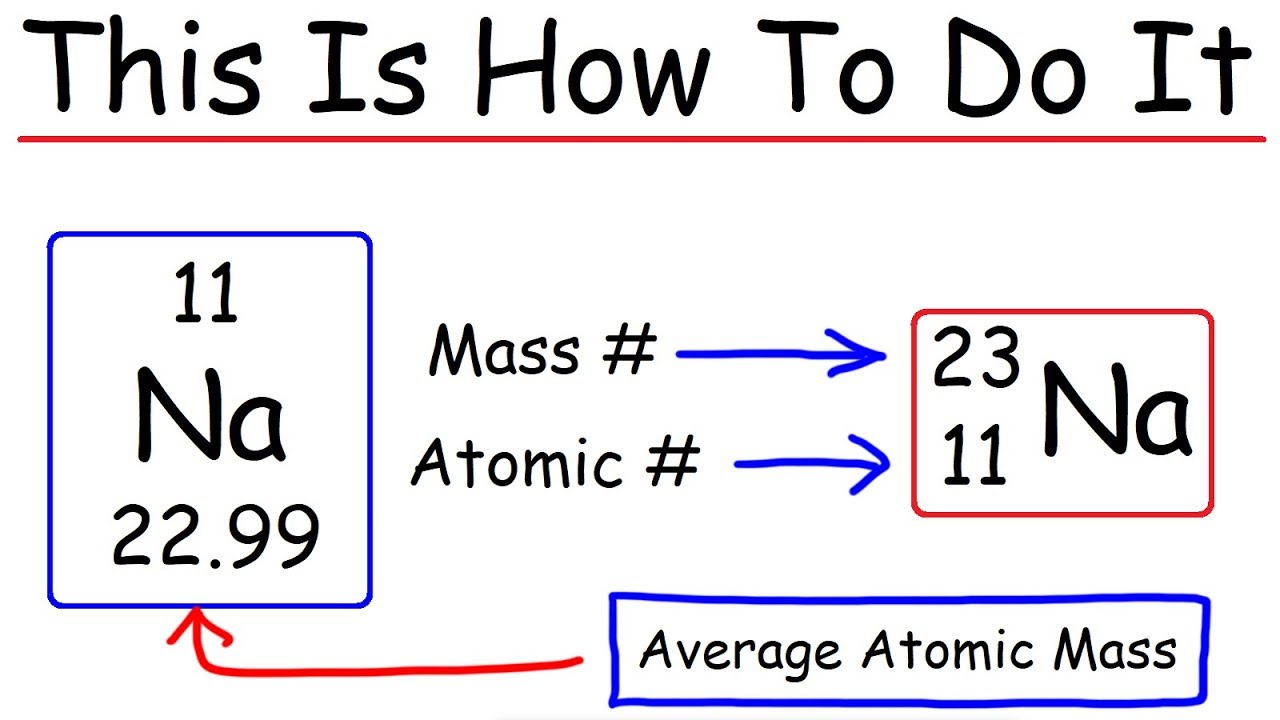
So to get the number of electrons, you must add the size of charge to the atomic or proton number.
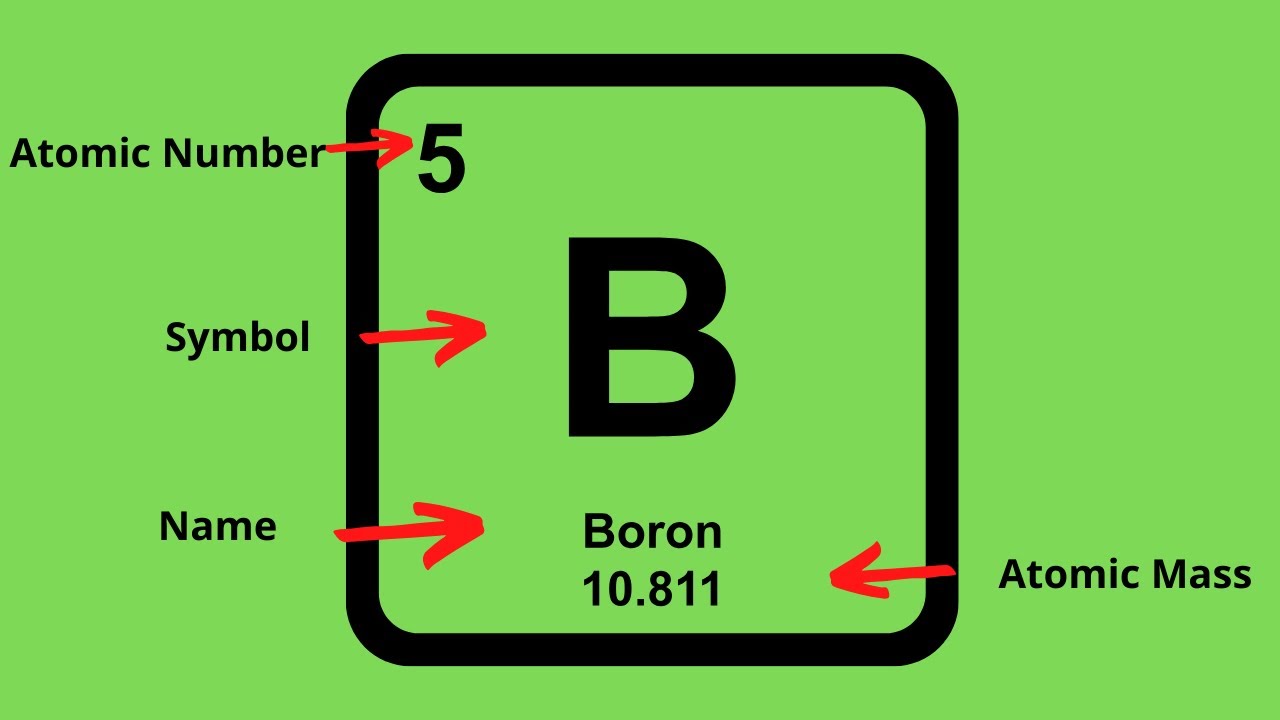
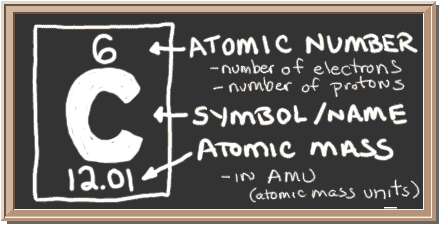
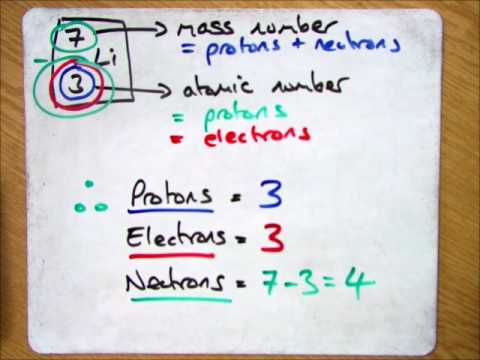
How to find out electrons. Thomson discovered the existence of electrons through cathode ray examination. Number of electrons = number of protons = atomic number; How to calculate the number of electrons?
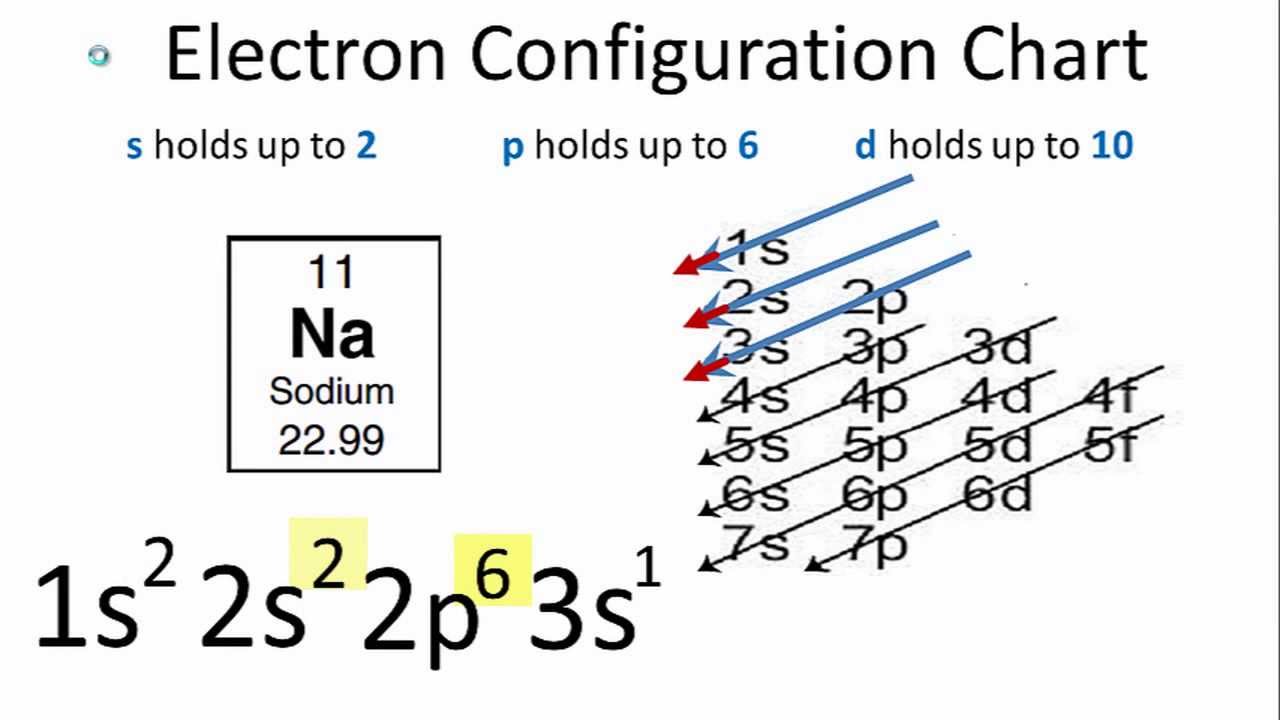
Since the atom is neutral, the number of electrons is equal to the proton. So the electron configuration is 1s^2, 2s^2, 2p^6, 3s^2, 3p^3 (the exponents add up tp 15). The smallest of the permanent core particles of an atom is the electron.
This is why we have the 2 n 2 rule. Mass number = sum of protons and neutrons; So from figure 3, the number of electrons for chloride ion is 17 + 1 = 18.
There are as many orbitals in the n shell as the sum of the first mathrm n odd numbers:. The number of protons in the nucleus of the atom is equal to the. You can find valence electrons with a shortcut using the periodic table, but it’s good to only do that after you understand why the shortcut works, and to do that you have to draw out the full.
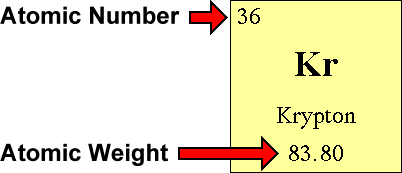
The atomic number is the number of. If the ion is negatively charged, the number of electrons is found by adding the charge number to the proton number. The number of electrons in a neutral atom is equal to the number of protons.
The 1 st subshell (p) can hold 3 orbitals, or 6 electrons. You can also find the core and valence electrons by determining or looking up the electron configurations of the main group elements. Number of protons = number of electrons = atomic number number of neutrons = mass.
The electron configuration of an atom is written with the help of subshell labels. These labels contain the shell number (given by the principal quantum number), the subshell name (given by.